The paper is available for download (PDF) in following languages:
English French German Indonesian Polish Portuguese Russian SwahiliWhat are nicotine pouches, and how are they made?
Nicotine pouches are thumbnail-sized sachets containing vegetable fibres infused with nicotine and a range of flavours. Placed in the mouth between the lip and gum, nicotine is absorbed through the oral mucous membrane. This Briefing Paper provides an introduction to the nicotine pouch category and assesses the potential for these products to contribute to tobacco harm reduction.
Nicotine pouches were first produced in the early 2000s. Due to their similar appearance and method of use, nicotine pouches are sometimes confused with Swedish snus, another safer nicotine product (SNP) that is placed between the lip and gum.[i] But while Swedish snus contains tobacco, nicotine pouches do not contain any raw or processed tobacco leaves. The nicotine used in pouches may be synthetic, or extracted from tobacco plants.
Nicotine pouches from major companies are normally marketed by strength, for example: low (1.5mg), regular (3mg), strong (6mg) and extra-strong (9mg).[ii] However, they can vary in nicotine content, with one study that assessed different products finding this ranged from 1.79 mg to 47.5mg per pouch.[iii] Well-known brands include Velo (manufactured by BAT), on! (Altria) and ZYN (Swedish Match).
Are nicotine pouches a safer alternative to high-risk tobacco products?
The health impacts of smoking are now well understood to be caused by the combustion of tobacco which produces a cocktail of harmful chemicals found in smoke. But since nicotine pouches contain no tobacco, and require no combustion, their use avoids many of the risks associated with smoking that result from the burning of tobacco.
Extensive scientific research has been conducted on nicotine. It is a comparatively low-risk drug that does not cause any of the severe illnesses associated with high-risk tobacco products like cigarettes. It is not carcinogenic and has been deemed “no more harmful to health than caffeine” by the Royal Society for Public Health in the UK.[iv] It is also on the World Health Organization’s list of Essential Medicines, and medical practitioners have been prescribing nicotine patches, lozenges and gums to people trying to quit smoking for decades.
But while nicotine is a very well-understood chemical, nicotine pouches are still quite a new product. There is therefore a smaller body of evidence on their relative safety compared to the growing scientific literature analysing other SNP such as nicotine vapes (e-cigarettes) and snus. However, research on some of these similar products can help provide insights into the potential impact of using nicotine pouches.
Oral products in the nicotine replacement therapy (NRT) category are a useful comparator, providing a good indication of the relative safety of nicotine pouches. Nicotine lozenges are relatively similar products to nicotine pouches; neither contain tobacco and both involve the oral absorption of nicotine, which is their main active component. The US Centers for Disease Control and Prevention (CDC) has stated that nicotine replacement therapies in the form of gums, lozenges and patches are not carcinogenic, and has recommended them as alternatives to smoking. The CDC also asserts that NRT are “much less addictive than cigarettes”, adding that “NRT delivers less nicotine to your brain and much more slowly”.[v]
Swedish snus is another comparable product to nicotine pouches, with a similar mode of use. Snus has a well-established history dating back 200 years and several decades of research on the modern version of the product have proven it to be both a safer alternative to smoking and a successful smoking cessation product.[vi] High levels of snus use in Sweden and Norway have been associated with low smoking rates in these countries - 23% of adult Swedish men use snus,[vii] and Sweden is the only country in the European Union to have achieved ‘smoke-free status’, meaning the adult smoking prevalence is under 5%. Snus has significantly lower levels of key toxicants than are found in cigarette smoke, including tobacco-specific nitrosamines (TSNAs). These toxicants are carcinogens that are linked to cancers of the lung, oral cavity, liver and oesophagus, and research has found there to be no association between Swedish snus use and oral cancer.[viii][ix]
The US Food and Drug Administration has authorised the marketing of eight snus products sold under the 'General' brand name stating that “using General Snus instead of cigarettes puts you at a lower risk of mouth cancer, heart disease, lung cancer, stroke, emphysema, and chronic bronchitis”.[x]
When compared to other nicotine delivery systems, nicotine pouches are considered to pose an even lower risk to health than snus, as shown in this diagram below.
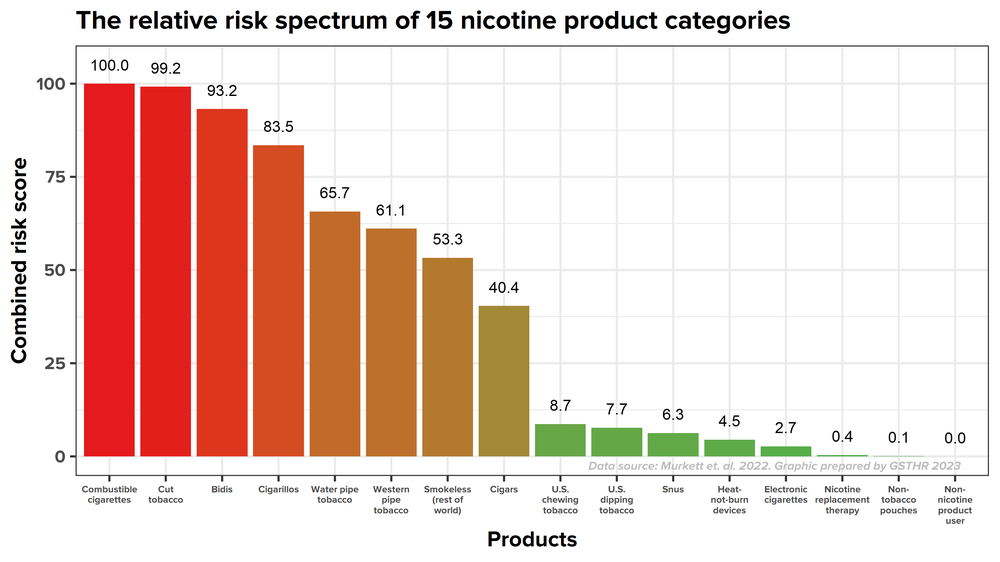
The relative risk hierarchy of the 15 categories of nicotine products. Adapted from Murkett et al [xi] with permission.
This is supported by research conducted into the relative levels of TSNAs found in both products. In a study comparing the quantities of toxicants found in Swedish snus and nicotine pouches, it was found that the levels of several TSNAs were measurably lower in nicotine pouches than in Swedish snus. The TSNA concentrations found in nicotine pouches were comparable to those found in oral NRT.[xii]
The body of evidence examining nicotine pouch safety is still growing, but these preliminary findings indicate that nicotine pouches are a significantly safer alternative to smoking.
Could nicotine pouches provide a safer alternative for users of high-risk tobacco products?
Nicotine pouches have the potential to make a significant contribution to tobacco harm reduction, which offers people who use high-risk tobacco products, like cigarettes and some oral tobaccos, the chance to switch to safer nicotine products that pose significantly fewer risks to their health. This is particularly the case for users of Asian-type smokeless tobacco products, which can also contain slaked lime (calcium hydroxide) and other hazardous ingredients. Smokeless tobacco refers to a range oftobacco-containing products that are consumed mainly by chewing or being kept in the mouth, though some can also be sniffed. Common types of such Asian-style smokeless tobacco include betel quid, paan or gutkha, all of which are oral products.
These smokeless tobacco products are used by more than 300 million people worldwide.[xiii] Posing a high risk to health, they contain relatively high levels of carcinogenic and toxic compounds and increase the risk of oral, oesophageal, and pancreatic cancers. (But these Asian-style smokeless tobacco products should not be confused with US smokeless tobacco products, including chewing tobacco and American moist snuff, which are associated with minimal risk for oral cancer.)[xiv] The countries that experience the greatest disease burden are those in South and Southeast Asia where Asian-style smokeless tobacco consumption is highest.
Nicotine pouches have the potential to replace these risky smokeless tobacco products, offering a safer alternative that poses significantly fewer risks to individual health. Substitution of risky smokeless tobacco with nicotine pouches could help reduce rates of non-communicable disease in numerous low-and middle-income countries (LMIC) where smokeless tobacco use is prevalent.
Nicotine pouches also provide a safer alternative to combustible cigarettes. Eighty percent of the world’s smokers live in LMIC, so nicotine pouches could be particularly effective in reducing the harms of smoking in countries where the cost and complexity of other SNPs may prove problematic.
Nicotine vapes and heated tobacco products require consumers to invest upfront in devices, which need electricity to charge. This may be unaffordable or impractical for consumers in LMIC. By contrast, nicotine pouches are relatively low-cost, low-tech and easy to use. They have a low environmental impact, with no batteries or other electronic components, and minimal packaging. As their use has no impact on bystanders, people can also use nicotine pouches in smoke-free environments where combustible products and vaping products may be banned.
Nicotine pouches are relatively simple and inexpensive to manufacture. In comparison with nicotine vapes and heated tobacco products, they do not require manufacturers to invest substantially in research and development. There are also currently few regulatory barriers to market entry in most countries.
How popular are nicotine pouches and who is using them?
Nicotine pouches are not as well known or widely used as other novel nicotine products, such as nicotine vapes. A 2022 survey conducted by Action on Smoking and Health, found that 4.6% of the population of Great Britain had tried nicotine pouches at some point, while just 0.7% of the population continue to use nicotine pouches. Meanwhile, 2.4% of the people who smoke are current users of nicotine pouches.[xv]
Nicotine pouches have seen their market size substantially grow in recent years. As of 2021, the nicotine pouch market had a global market size of 1.5bn USD.[xvi]
In the USA, the nicotine pouch market grew by just over 100% between 2020 and 2021.[xvii] At time of writing, the USA has the largest nicotine pouch market of any country (for which data have been made available). The USA market is currently dominated by ZYN (Swedish Match), which accounted for 58.8% of nicotine pouch unit sales in the period 2019–2022, with on! (Altria) and Velo (BAT) following behind with 24.6% and 12.1% of total unit sales respectively.[xviii] In the UK, the nicotine pouch market grew by 12% between 2020 and 2021.
The growing nicotine pouch market demonstrates a rise in interest in oral nicotine products among consumers. A 2021 study of current smokers in the USA found that 5.6% of those interviewed had tried nicotine pouches, with 16.8% of the group of smokers expressing an interest in trying nicotine pouches.[xix] A 2021 study of young adults who had never used nicotine pouches found that interest in nicotine pouches was highest among users of combustible tobacco, non-combustible tobacco and e-cigarettes.[xx] There was a lower, but not negligible, interest in nicotine pouches among non-users of any tobacco product. Although use of nicotine pouches by people who have never smoked appears to take place, there is initial data to suggest that this group makes up a minority of nicotine pouch users, with the majority being former or current users of cigarettes, smokeless tobacco or reduced-risk nicotine products. For example, a UK survey looking at nicotine pouch use during 2020-2021 found nicotine pouch use to be highest among current smokers and former smokers, rather than among never-smokers.[xxi] In a survey of ZYN nicotine pouch consumers in the USA, roughly 4% of ZYN consumers were never-smokers, with the majority of ZYN consumers being current or former smokers or users of smokeless tobacco.[xxii]
How are nicotine pouches regulated around the world?
Regulatory restrictions on nicotine pouches vary significantly globally at the time of writing. For example, in the European Union there is no harmonised regulation yet for the category and the different Member States treat nicotine pouches differently. Some countries, such as Sweden,[xxiii] Denmark,[xxiv] Hungary,[xxv] Czech Republic and Slovenia,[xxvi] have so far implemented specific regulation for nicotine pouches, while others have imposed taxation on the product (Sweden, Denmark, Hungary, Estonia and Latvia).[xxvii][xxviii][xxix] Finland only allows pouches as an NRT product,[xxx] while Belgium and the Netherlands are both currently deliberating banning nicotine pouches.[xxxi][xxxii] At this point in time, nicotine pouches are regarded as food products in both the Netherlands and Germany, effectively banning their presence on the market (as food products cannot contain nicotine).[xxxiii] However, the majority of EU Member States regulate nicotine pouches as a consumer product.
A report by the National Institute of Public Health and the Environment in the Netherlands, has recommended the implementation of EU-wide restrictions on the sale of nicotine pouches to minors, a ban on cross-border sales and restrictions on nicotine pouch flavours.[xxxiv] Reports indicate that the European Union, both through the Tobacco Products Directive and the Tax Excise Directive, will seek to introduce coherent regulation and tax restrictions for novel nicotine products, including nicotine pouches.[xxxv] The European Commission, in advice given to the European Parliament on the application of the Tobacco Products Directive, has highlighted nicotine pouches as an issue to be specifically addressed, due to the lack of EU-wide regulation of these products.[xxxvi]
The distorted regulatory situation across the European Union’s Member States is expected to change once legislation is harmonised across the European Union’s internal market. This regulatory picture is similar for European countries outside of the European Union.
In the United Kingdom, nicotine pouches are classed as consumer goods,[xxxvii] and therefore do not fall under tobacco product or medicinal product regulations. There is therefore no maximum nicotine strength requirement, and there are no restrictions on the advertising of nicotine pouches.[xxxviii] When the Khan Review, an independent report examining the UK government’s aim to become smoke-free, was published in June 2022, no specific recommendations about nicotine pouches were made.[xxxix] However, the Review did suggest that “instead [of legalising snus], the government must facilitate access to the various already available safer alternative nicotine products such as nicotine pouches (a tobacco-free equivalent of snus), maximising their value to help smokers to quit, without creating new risks to young people.” The New Nicotine Alliance, a UK-based consumer advocacy organisation, has called for nicotine pouches to be regulated in a similar manner to Slovakia, with bespoke legislation restricting maximum nicotine content.[xl]
In Norway, nicotine pouch products must be approved by the Health Directorate before they can be placed on the market. So far, no manufacturer has been granted authorisation to do this.[xli] However, a similar product to nicotine pouches called white snus (a type of snus containing mostly vegetable fibre infused with nicotine, with a small amount of added tobacco) is legally available for purchase in Norway as it is covered under existing legislation permitting the sale of snus.
Switzerland has introduced nicotine pouches in its recently adopted tobacco legislation, which comes into force in 2023. Under this legislation, nicotine pouch products must be accompanied by health warnings.[xlii]
Iceland has recently regulated nicotine pouches in its tobacco law including introducing a maximum nicotine content of 20mg/pouch.[xliii]
A ban on nicotine pouches in Kenya came into force in 2020, but has since been overturned, with sales of Velo nicotine pouches permitted under the Tobacco Control Act.[xliv][xlv]
In the USA, nicotine pouches are regulated as tobacco products under the Family Smoking Prevention and Tobacco Control Act. Premarket authorisation for nicotine pouch products must be obtained, and product packaging must feature health warning labels.[xlvi][xlvii]
Meanwhile, Australia has banned nicotine pouches along with all other novel nicotine products including nicotine vapes and heated tobacco products. The only exemptions are if these products are provided to individuals on prescription, as with nicotine vapes.[xlviii] Likewise, New Zealand has banned nicotine pouches,[xlix] while the Philippines regulate nicotine pouches as tobacco products.[l]
Conclusion
Nicotine pouches are a relatively new safer nicotine product which can provide users of high-risk tobacco with an alternative that poses fewer risks to their health. They are low-tech and have a low environmental impact as they contain no batteries or other electronic components. The rapid growth of the nicotine pouch market, already worth 1.5bn USD globally, shows users of high-risk tobacco find these products acceptable and are switching to them. More scientific research is needed to provide further evidence of their reduced-risk status, but it is already clear they offer the potential to save many lives in LMIC where high-risk oral tobacco use is prevalent.
About us: Knowledge·Action·Change (K·A·C) promotes harm reduction as a key public health strategy grounded in human rights. The team has over forty years of experience of harm reduction work in drug use, HIV, smoking, sexual health, and prisons. K·A·C runs the Global State of Tobacco Harm Reduction (GSTHR) which maps the development of tobacco harm reduction and the use, availability and regulatory responses to safer nicotine products, as well as smoking prevalence and related mortality, in over 200 countries and regions around the world. For all publications and live data, visit https://gsthr.org
Our funding: The GSTHR project is produced with the help of a grant from the Foundation for a Smoke-Free World, a US non profit 501(c)(3), independent global organization. The project and its outputs are, under the terms of the grant agreement, editorially independent of the Foundation.
[i] GSTHR. (2022). An introduction to snus (GSTHR Briefing Papers). Global State of Tobacco Harm Reduction. https://gsthr.org/briefing-papers/an-introduction-to-snus/.
[ii] What Strength of ZYN is Right for You? (2022, September 5). Zyn UK. https://uk.zyn.com/blog/our-products/what-strength-of-zyn-is-right-for-you/.
[iii] Mallock, N., Schulz, T., Malke, S., Dreiack, N., Laux, P., & Luch, A. (2022). Levels of nicotine and tobacco-specific nitrosamines in oral nicotine pouches. Tobacco Control. https://doi.org/10.1136/tc-2022-057280.
[iv] Royal Society of Public Health. (2015a). Stopping smoking by using other sources of nicotine. https://www.rsph.org.uk/resourceLibrary/stopping-smoking-by-using-other-sources-of-nicotine.html; (2015b, August 13). Nicotine “no more harmful to health than caffeine”. https://www.rsph.org.uk/about-us/news/nicotine--no-more-harmful-to-health-than-caffeine-.html?s=03.
[v] Quit smoking medicines are much safer than smoking | Quit Smoking | Tips From Former Smokers. (2022, November 28). Centers for Disease Control and Prevention. https://www.cdc.gov/tobacco/campaign/tips/quit-smoking/quit-smoking-medications/3-reasons-to-use-medicines-when-you-quit/quit-medicines-are-safer-than-smoking/index.html.
[vi] Gartner, C. E., Hall, W. D., Vos, T., Bertram, M. Y., Wallace, A. L., & Lim, S. S. (2007). Assessment of Swedish snus for tobacco harm reduction: An epidemiological modelling study. The Lancet, 369(9578), 2010–2014. https://doi.org/10.1016/S0140-6736(07)60677-1.
[vii] Statistics Sweden. (n.d.). Tobacco habits by indicator, study domain and sex. Percentage and estimated numbers in thousands. Year 2008-2009—2021-2021. Statistikdatabasen. Retrieved 27 September 2022, from http://www.statistikdatabasen.scb.se/pxweb/en/ssd/START__LE__LE0101__LE0101H/LE01012021H06/.
[viii] Araghi, M., Galanti, M. R., Lundberg, M., Liu, Z., Ye, W., Lager, A., Engström, G., Alfredsson, L., Knutsson, A., Norberg, M., Wennberg, P., Lagerros, Y. T., Bellocco, R., Pedersen, N. L., Östergren, P.-O., & Magnusson, C. (2021). No association between moist oral snuff (snus) use and oral cancer: Pooled analysis of nine prospective observational studies. Scandinavian Journal of Public Health, 49(8), 833–840. https://doi.org/10.1177/1403494820919572.
[ix] Lee, P. N. (2011). Summary of the epidemiological evidence relating snus to health. Regulatory Toxicology and Pharmacology: RTP, 59(2), 197–214. https://doi.org/10.1016/j.yrtph.2010.12.002.
[x] US Food & Drug Administration. (2020, March 24). FDA grants first-ever modified risk orders to eight smokeless tobacco products. FDA. https://www.fda.gov/news-events/press-announcements/fda-grants-first-ever-modified-risk-orders-eight-smokeless-tobacco-products.
[xi] Murkett, R., Rugh, M., & Ding, B. (2022). Nicotine products relative risk assessment: An updated systematic review and meta-analysis (9:1225). F1000Research. https://doi.org/10.12688/f1000research.26762.2.
[xii] Azzopardi, D., Liu, C., & Murphy, J. (2022). Chemical characterization of tobacco-free “modern” oral nicotine pouches and their position on the toxicant and risk continuums. Drug and Chemical Toxicology, 45(5), 2246–2254. https://doi.org/10.1080/01480545.2021.1925691.
[xiii] Siddiqi, K., Husain, S., Vidyasagaran, A., Readshaw, A., Mishu, M. P., & Sheikh, A. (2020). Global burden of disease due to smokeless tobacco consumption in adults: An updated analysis of data from 127 countries. BMC Medicine, 18(1), 222. https://doi.org/10.1186/s12916-020-01677-9.
[xiv] Rodu, B., & Jansson, C. (2004). Smokeless tobacco and oral cancer: A review of the risks and determinants. Critical Reviews in Oral Biology and Medicine: An Official Publication of the American Association of Oral Biologists, 15(5), 252–263. https://doi.org/10.1177/154411130401500502.
[xv] ASH. (2022). Awareness and use of nicotine pouches. Action on Smoking and Health. https://ash.org.uk/resources/view/awareness-and-use-of-nicotine-pouches.
[xvi] GVR. (n.d.). Nicotine Pouches Market Size, Share & Trends Analysis Report By Product (Tobacco-derived, Synthetic), By Flavor (Original/Unflavored, Flavored), By Strength, By Distribution Channel, By Region, And Segment Forecasts, 2022—2030. Grand View Research. Retrieved 23 March 2023, from https://www.grandviewresearch.com/industry-analysis/nicotine-pouches-market-report.
[xvii] Tobacco free oral nicotine market size in USD by country up to 2021, data sourced from Euromonitor, with processing by GSTHR.
[xvii] Majmundar, A., Okitondo, C., Xue, A., Asare, S., Bandi, P., & Nargis, N. (2022). Nicotine Pouch Sales Trends in the US by Volume and Nicotine Concentration Levels From 2019 to 2022. JAMA Network Open, 5(11), e2242235. https://doi.org/10.1001/jamanetworkopen.2022.42235.
[xix] Hrywna, M., Gonsalves, N. J., Delnevo, C. D., & Wackowski, O. A. (2022). Nicotine pouch product awareness, interest and ever use among US adults who smoke, 2021. Tobacco Control. https://doi.org/10.1136/tobaccocontrol-2021-057156.
[xx] Vogel, E. A., Barrington-Trimis, J. L., Kechter, A., Tackett, A. P., Liu, F., Sussman, S., Lerman, C., Unger, J. B., Hughes Halbert, C., Chaffee, B. W., & Leventhal, A. M. (2022). Differences in Young Adults’ Perceptions of and Willingness to Use Nicotine Pouches by Tobacco Use Status. International Journal of Environmental Research and Public Health, 19(5), 2685. https://doi.org/10.3390/ijerph19052685.
[xxi] Tattan-Birch, H., Jackson, S. E., Dockrell, M., & Brown, J. (2022). Tobacco-free Nicotine Pouch Use in Great Britain: A Representative Population Survey 2020–2021. Nicotine & Tobacco Research, 24(9), 1509–1512. https://doi.org/10.1093/ntr/ntac099.
[xxii] Plurphanswat, N., Hughes, J. R., Fagerström, K., & Rodu, B. (2020). Initial Information on a Novel Nicotine Product. The American Journal on Addictions, 29(4), 279–286. https://doi.org/10.1111/ajad.13020.
[xxiii] Tobacco free nicotine products. (2022, October 14). The Public Health Agency of Sweden. https://www.folkhalsomyndigheten.se/the-public-health-agency-of-sweden/living-conditions-and-lifestyle/andtg/rules/tobacco-free-nicotine-products/.
[xxiv] Klitgaard, M. B., Jarlstrup, N. S., Lund, L., Brink, A.-L., Knudsen, A., Christensen, A. I., & Bast, L. S. (2022). Evaluating the Effects of Denmark’s New Tobacco Control Act on Young People’s Use of Nicotine Products: A Study Protocol of the §SMOKE Study. International Journal of Environmental Research and Public Health, 19(19), 12782. https://doi.org/10.3390/ijerph191912782.
[xxv] Riudalbas, S. (2022, March 30). Regulatory report: Hungary, March 2022. TobaccoIntelligence. https://tobaccointelligence.com/regulatory-report-hungary-hnb-oral-tobacco-and-nicotine-pouches-march-2022/.
[xxvi] TobaccoIntelligence. (2023, January 25). Lawmakers in the Czech Republic look to regulate nicotine pouches in two acts. TobaccoIntelligence. https://tobaccointelligence.com/lawmakers-in-the-czech-republic-look-to-regulate-nicotine-pouches-in-two-acts/.
[xxvii] Bill to an Act amending the Act on Various Consumption Taxes and the Tax Collection Act (Introduction of tax on nicotine products and aggregation of tax rates on smokeless tobacco, etc.), no. 2020–9326, The Danish Ministry of Taxation (Skatteministeriet) (2021). https://www.google.com/url?sa=t&rct=j&q=&esrc=s&source=web&cd=&ved=2ahUKEwjCmaesw6v9AhVIPcAKHUoQBiYQFnoECAwQAQ&url=https%3A%2F%2Fec.europa.eu%2Fgrowth%2Ftools-databases%2Ftris%2Findex.cfm%2Fsk%2Fsearch%2F%3Ftrisaction%3Dsearch.detail%26year%3D2021%26num%3D703%26dLang%3DEN&usg=AOvVaw1gGX02iZ2drKrMRaK07CbW.
[xxviii] Sweden: New Tax on Electronic Cigarettes and Other Non-Tobacco Nicotine Products Takes Effect. (2018). [Web page]. Library of Congress, Washington, D.C. 20540 USA. https://www.loc.gov/item/global-legal-monitor/2018-07-26/sweden-new-tax-on-electronic-cigarettes-and-other-non-tobacco-nicotine-products-takes-effect/.
[xxix] Pauwels, C., Bakker-’t Hart, I., Hegger, I., Bil, W., Bos, P., & Talhout, R. (2021). Nicotineproducten zonder tabak voor recreatief gebruik. Rijksinstituut voor Volksgezondheid en Milieu. https://doi.org/10.21945/RIVM-2020-0152.
[xxx] Salokannel, M., & Ollila, E. (2021). Snus and snus-like nicotine products moving across Nordic borders: Can laws protect young people? Nordic Studies on Alcohol and Drugs, 38(6), 540–554. https://doi.org/10.1177/1455072521995704.
[xxxi] Royal Decree prohibiting the manufacture and placing on the market of certain similar products, European Commission, Internal Market, Industry, Entrepreneurship and SMEs (2022). https://ec.europa.eu/growth/tools-databases/tris/en/search/?trisaction=search.detail&year=2022&num=417.
[xxxii] Dutch Danger. (2022, December 23). European Tobacco Harm Reduction Advocates (ETHRA). https://ethra.co/news/121-dutch-danger.
[xxxiii] Dutch Danger, 2022.
[xxxiv] Pauwels, Bakker-’t Hart, Hegger, Bil, Bos, & Talhout, 2021.
[xxxv] Michalopoulos, S. (2022, October 12). EU-wide excise tax on novel tobacco products: A tough equation to solve. Euractiv. https://www.euractiv.com/section/health-consumers/news/eu-wide-excise-tax-on-novel-tobacco-products-a-tough-equation-to-solve/.
[xxxvi] Report from the Commission to the European Parliament, the Council, the European Economic and Social Committee and the Committee of the Regions on the application of Directive 2014/40/EU concerning the manufacture, presentation and sale of tobacco and related products (COM(2021) 249 final). (2021). European Commission. https://eur-lex.europa.eu/legal-content/EN/TXT/HTML/?uri=CELEX:52021DC0249&from=EN.
[xxxvii] ASH, 2022.
[xxxviii] Action on Smoking and Health (ASH). (2021). UK Product Safety Review: Call for Evidence Response from Action on Smoking and Health and SPECTRUM. https://ash.org.uk/uploads/UK-Product-Safety-Review_ASH_SPECTRUM_FINAL.pdf?v=1660663875.
[xxxix] Khan, J. (2022, June 9). The Khan review: Making smoking obsolete. Department of Health, UK. https://www.gov.uk/government/publications/the-khan-review-making-smoking-obsolete.
[xl] Ahmed, I. (2022, November 27). The NNA welcomes calls for proportionate regulation of nicotine pouches. New Nicotine Alliance UK. https://nnalliance.org/blog/380-the-nna-welcomes-calls-for-proportionate-regulation-of-nicotine-pouches.
[xli] Dawson, F. (2022, February 9). Changes to Norwegian rules unlikely to have much impact on the market. TobaccoIntelligence. https://tobaccointelligence.com/changes-to-norwegian-rules-unlikely-to-have-much-impact-on-the-market/.
[xlii] Rubio, D. P. (2021, November 4). Switzerland moves ahead with bill to regulate heated and oral tobacco. TobaccoIntelligence. https://tobaccointelligence.com/switzerland-moves-ahead-with-bill-to-regulate-heated-and-oral-tobacco/.
[xliii] Lorenzo, A. di. (2022, September 14). Iceland’s new regulations for pouches include 2% cap on nicotine. TobaccoIntelligence. https://tobaccointelligence.com/icelands-new-regulations-for-pouches-include-2-cap-on-nicotine-content/.
[xliv] British American Tobacco (BAT). (2023). New Category Acceleration Drives Profitability Forward To 2024 [Press release]. https://www.bat.com/group/sites/UK__9D9KCY.nsf/vwPagesWebLive/DOCNUNN9/$file/FY_2022_Announcement.pdf.
[xlv] Aloo, H. (2022, August 31). BAT reverses ban to relaunch nicotine pouches in Kenya. The Africa Report. https://www.theafricareport.com/234861/bat-reverses-ban-to-relaunch-nicotine-pouches-in-kenya/.
[xlvi] Pauwels, Bakker-’t Hart, Hegger, Bil, Bos, & Talhout, 2021.
[xlvii] Center for Tobacco Products. (2022). Requirements for Products Made with Non-Tobacco Nicotine Take Effect April 14. US Food & Drug Administration (FDA). https://www.fda.gov/tobacco-products/ctp-newsroom/requirements-products-made-non-tobacco-nicotine-take-effect-april-14.
[xlviii] Pauwels, Bakker-’t Hart, Hegger, Bil, Bos, & Talhout, 2021.
[xlix] Smokefree Environments and Regulated Products (Vaping) Amendment Bill, Government Bill – New Zealand Legislation, no. 222–2 (2020). https://legislation.govt.nz/bill/government/2020/0222/latest/whole.html#d16822133e2.
[l] Carrasco, E. (2020, October 7). Regulatory report: Philippines – heated tobacco and modern oral tobacco, July 2020. TobaccoIntelligence. https://tobaccointelligence.com/regulatory-report-philippines-heated-tobacco-and-modern-oral-tobacco/.